m6A-centered Crosstalk Information
Mechanism of Crosstalk between m6A Modification and Epigenetic Regulation
| Crosstalk ID |
M6ACROT05830
|
[1] | |||
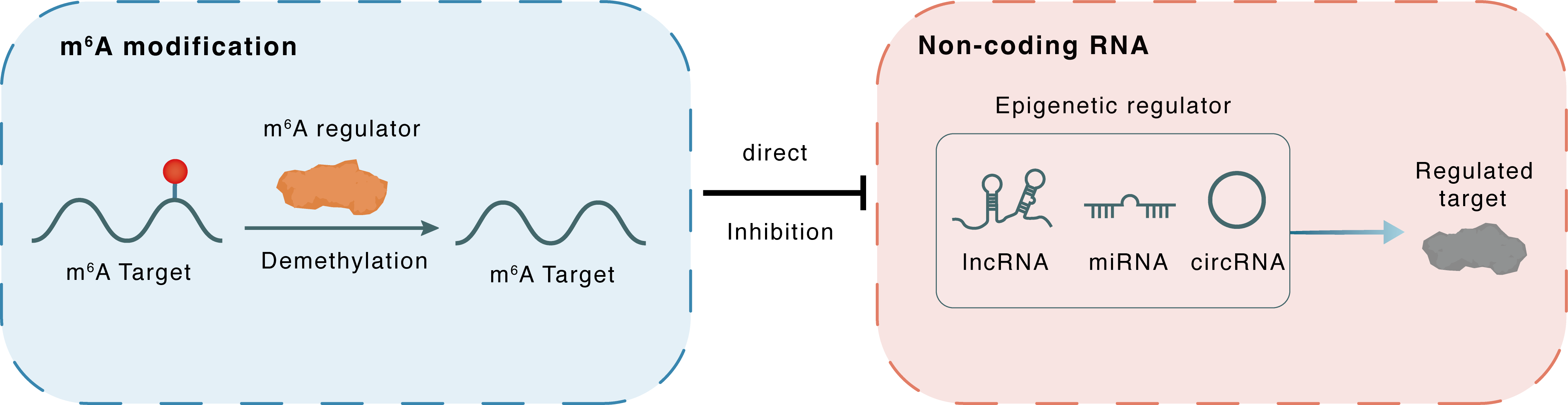 m6A modification
MEG3
MEG3
FTO
Demethylation
m6A modification
MEG3
MEG3
FTO
Demethylation
 : m6A sites
Direct
Inhibition
Non-coding RNA
MEG3
EZH2
lncRNA miRNA circRNA : m6A sites
Direct
Inhibition
Non-coding RNA
MEG3
EZH2
lncRNA miRNA circRNA
|
|||||
| m6A Modification: | |||||
|---|---|---|---|---|---|
| m6A Regulator | Fat mass and obesity-associated protein (FTO) | ERASER | |||
| m6A Target | Maternally expressed 3 (MEG3) | ||||
| Epigenetic Regulation that have Cross-talk with This m6A Modification: | |||||
| Epigenetic Regulation Type | Non-coding RNA (ncRNA) | ||||
| Epigenetic Regulator | Maternally expressed 3 (MEG3) | LncRNA | View Details | ||
| Regulated Target | Histone-lysine N-methyltransferase EZH2 (EZH2) | View Details | |||
| Downstream Gene | TGFB1 | View Details | |||
| Crosstalk Relationship | m6A → ncRNA | Inhibition | |||
| Crosstalk Mechanism | m6A regulators directly modulate the functionality of ncRNAs through specific targeting ncRNA | ||||
| Crosstalk Summary | FTO regulates the Maternally expressed 3 (MEG3)-TGFB1 signalling pathway in a YTHDC1 dependent manner by binding simultaneously with the Histone-lysine N-methyltransferase EZH2 (EZH2), thereby suppressing trophoblast invasion and proliferation in URSA trophoblast cells. These findings provide new insights for the treatment of URSA. | ||||
| Responsed Disease | Abortion | ICD-11: JA00.0 | |||
| Cell Process | Cell proliferation | ||||
| In-vivo Model | Sexually mature ICR mice, aged at least 10 weeks, were paired as one female and one male per cage to obtain pregnancies. The time of the vaginal plug sighting was denoted as day 0.5 of the pregnancy. The mice were divided into an FTO protein group and control group, with 10 pairs of mice in each group. Starting on day 0.5 of the pregnancy, mice in the FTO protein group were intraperitoneally injected daily with FTO protein (3.5 ng/kg), whereas those in the control group had daily intraperitoneal injections of equal volumes of saline. Female mice were euthanised on day 13.5 of the pregnancy (day 12.5-14.5), and the total numbers of normal and resorbed embryos were determined, along with the rate of embryo resorption. | ||||
Full List of Potential Compound(s) Related to This m6A-centered Crosstalk
| Histone-lysine N-methyltransferase EZH2 (EZH2) | 74 Compound(s) Regulating the Target | Click to Show/Hide the Full List | ||
| Tazemetostat | Approved | [2] | ||
| Synonyms |
EPZ-6438; 1403254-99-8; EPZ6438; UNII-Q40W93WPE1; N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-5-(ethyl(tetrahydro-2H-pyran-4-yl)amino)-4-methyl-4'-(morpholinomethyl)-[1,1'-biphenyl]-3-carboxamide; Q40W93WPE1; EPZ 6438; E7438; (1,1'-Biphenyl)-3-carboxamide, N-((1,2-dihydro-4,6-dimethyl-2-oxo-3-pyridinyl)methyl)-5-(ethyl(tetrahydro-2H-pyran-4-yl)amino)-4-methyl-4'-(4-morpholinylmethyl)-
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| Activity | Ki = 2.5 nM | |||
| External Link | ||||
| DS-3201b | Phase 2 | [3] | ||
| Synonyms |
Valemetostat; 1809336-39-7; UNII-60RD0234VE; 60RD0234VE; 1809336-39-7 (free base); DS-3201; (2R)-7-chloro-2-[trans-4-(dimethylamino)cyclohexyl]-N-[(4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl]-2,4-dimethyl-1,3-benzodioxole-5-carboxamide; Valemetostat 2HCl; Valemetostat [INN]; Valemetostat (DS-3201); CHEMBL4597193; EZH1/2 inhibitor DS-3201; SCHEMBL18393626; SCHEMBL18393627; SCHEMBL18639210; EX-A3423; DS3201; NSC813381; s8926; NSC-813381; HY-109108; CS-0039740; D11551
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| CPI-1205 | Phase 1/2 | [4] | ||
| Synonyms |
HPODOLXTMDHLLC-QGZVFWFLSA-N; 1621862-70-1; UNII-455J2479FY; CPI1205; CPI 1205; 455J2479FY; (R)-N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl-1-(1-(1-(2,2,2-trifluoroethyl)piperidin-4-yl)ethyl)-1H-indole-3-carboxamide; GTPL9115; SCHEMBL17329268; MolPort-044-560-382; KS-000006BA; EX-A1068; s8353; AKOS030628484; ZINC220982768; CS-7648; compound 13 [PMID: 27739677]; HY-100021; J3.556.402K; N-[(4-methoxy-6-methyl-2-oxo-1H-pyridin-3-yl)methyl]-2-methyl-1-[(1R)-1-[1-(2,2,2-trifluoroethyl)piperidin-4-
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| SHR2554 | Phase 1/2 | [5] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| CPI-0209 | Phase 1/2 | [6] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| GSK2816126 | Phase 1 | [7] | ||
| Synonyms |
GSK 126; GSK-126
Click to Show/Hide
|
|||
| MOA | Modulator | |||
| Activity | Ki = 0.5 nM | |||
| External Link | ||||
| PF-06821497 | Phase 1 | [8] | ||
| Synonyms |
UNII-S4L4MM20B6; S4L4MM20B6; CHEMBL4080228; PF06821497; 1844849-10-0; 5,8-dichloro-2-[(4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl]-7-[(R)-methoxy(oxetan-3-yl)methyl]-3,4-dihydroisoquinolin-1(2H)-one; SCHEMBL17330426; GTPL10516; BDBM50246967; NSC800019; DB14799; HY-101571A; NSC-800019; compound 23a [PMID: 29211475]; CS-0092626; Q29209799; 1(2H)-Isoquinolinone, 5,8-dichloro-2-((1,2-dihydro-4-methoxy-6-methyl-2-oxo-3-pyridinyl)methyl)-3,4-dihydro-7-((R)-methoxy-3-oxetanylmethyl)-; 5,8-dichloro-2-[(4-methoxy-6-methyl-2-oxo-1H-pyridin-3-yl)methyl]-7-[(R)-methoxy(oxetan-3-yl)methyl]-3,4-dihydroisoquinolin-1-one; CJD
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| DS-3201 | Phase 1 | [4] | ||
| Synonyms |
QTGYNKYZRZATJB-UHFFFAOYSA-N; 701899-63-0; 2-(2-(2-Methyl-1H-imidazol-5-yl)ethyl)isoindoline-1,3-dione; 2-(2-(2-Methyl-1H-imidazol-5-yl)-ethyl)isoindoline-1,3-dione; SCHEMBL7743461; MolPort-035-945-474; MolPort-035-690-353; ZINC98086069; AKOS024459002; AKOS024262663; MCULE-2708350770; FCH4077443; AK158834; AX8292467; ST2403812; 2-[2-(2-methyl-1H-imidazol-5-yl)ethyl]-2,3-dihydro-1H-isoindole-1,3-dione
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| HH2853 | Phase 1 | [9] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-33 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 100 nM | |||
| External Link | ||||
| PMID28394193-Compound-Figure3bI | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-35 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 10 nM | |||
| External Link | ||||
| PMID28394193-Compound-54 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-24 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 1000 nM | |||
| External Link | ||||
| PMID28394193-Compound-27 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 1000 nM | |||
| External Link | ||||
| PMID28394193-Compound-25 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 1000 nM | |||
| External Link | ||||
| PMID28394193-Compound-50 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 > 1 nM; IC50 < 2 nM; Ki > 0.01 nM; Ki < 0.04 nM | |||
| External Link | ||||
| PMID28394193-Compound-47 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 2 nM; Ki < 1 nM | |||
| External Link | ||||
| PMID28394193-Compound-21 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-41 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-53 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 80 nM | |||
| External Link | ||||
| PMID28394193-Compound-Figure5aVIII | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-38 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-51 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 > 1 nM; IC50 < 2 nM; Ki > 0.01 nM; Ki < 0.04 nM | |||
| External Link | ||||
| PMID28394193-Compound-31 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 20 nM | |||
| External Link | ||||
| PMID28394193-Compound-42 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-15 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-52 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 > 1 nM; IC50 < 2 nM; Ki > 0.01 nM; Ki < 0.04 nM | |||
| External Link | ||||
| PMID28394193-Compound-32 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 500 nM | |||
| External Link | ||||
| PMID28394193-Compound-23 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 1000 nM | |||
| External Link | ||||
| PMID28394193-Compound-29 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 100 nM | |||
| External Link | ||||
| PMID28394193-Compound-30 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 16 nM | |||
| External Link | ||||
| PMID28394193-Compound-39 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-49 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 > 1 nM; IC50 < 2 nM; Ki > 0.01 nM; Ki < 0.04 nM | |||
| External Link | ||||
| PMID28394193-Compound-43 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-40 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-Figure3bIII | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-36 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 316 nM | |||
| External Link | ||||
| PMID28394193-Compound-28 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 32 nM | |||
| External Link | ||||
| PMID28394193-Compound-22 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-18 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-16 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-44 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-20 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-19 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-37 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-Figure3bII | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-26 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 1000 nM | |||
| External Link | ||||
| PMID28394193-Compound-17 | Patented | [10] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-34 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 100 nM | |||
| External Link | ||||
| PMID28394193-Compound-56 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 0.93 nM | |||
| External Link | ||||
| PMID28394193-Compound-46 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 5 nM | |||
| External Link | ||||
| PMID28394193-Compound-57 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 6.45 nM | |||
| External Link | ||||
| PMID28394193-Compound-14 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 21900 nM | |||
| External Link | ||||
| PMID28394193-Compound-12 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 10 nM | |||
| External Link | ||||
| PMID26882240-Compound-1 | Patented | [11] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID28394193-Compound-55 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 8.13 nM | |||
| External Link | ||||
| PMID28394193-Compound-45 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 168000 nM | |||
| External Link | ||||
| PMID28394193-Compound-13 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 1470 nM | |||
| External Link | ||||
| EPZ005687 | Investigative | [12] | ||
| Synonyms |
EPZ-005687; EPZ 005687
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| EI1 | Investigative | [13] | ||
| Synonyms |
KB-145943
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| UNC1999 | Investigative | [14] | ||
| Synonyms |
UNC 1999; UNC-1999
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| MS1943 | Investigative | [5] | ||
| Synonyms |
2225938-17-8; SCHEMBL21271666; EX-A3962; s8918; HY-133129; CS-0112146; 6-(6-(4-(2-(2-((3r,5r,7r)-adamantan-1-yl)acetamido)ethyl)piperazin-1-yl)pyridin-3-yl)-N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-1H-indazole-4-carboxamide; 6-(6-(4-(2-(2-(Adamantan-1-yl)acetamido)ethyl)piperazin-1-yl)pyridin-3-yl)-N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-1H-indazole-4-carboxamide
Click to Show/Hide
|
|||
| MOA | Degrader | |||
| External Link | ||||
| GSK343 | Investigative | [15] | ||
| Synonyms |
compound 6 [PMID 24900432]; GSK 343
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| Activity | IC50 = 174 nM | |||
| External Link | ||||
| PMID28394193-Compound-11 | Patented | [16] | ||
| External Link | ||||
| PMID28394193-Compound-10 | Patented | [16] | ||
| External Link | ||||
| PMID28394193-Compound-14 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 21900 nM | |||
| External Link | ||||
| PMID28394193-Compound-12 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 < 10 nM | |||
| External Link | ||||
| PMID28394193-Compound-13 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 1470 nM | |||
| External Link | ||||
| PMID28394193-Compound-56 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 0.93 nM | |||
| External Link | ||||
| PMID28394193-Compound-46 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 5 nM | |||
| External Link | ||||
| PMID28394193-Compound-57 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 6.45 nM | |||
| External Link | ||||
| PMID28394193-Compound-55 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 8.13 nM | |||
| External Link | ||||
| PMID28394193-Compound-45 | Patented | [10] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 168000 nM | |||
| External Link | ||||
| Transforming growth factor beta 1 (TGFB1) | 10 Compound(s) Regulating the Target | Click to Show/Hide the Full List | ||
| Pirfenidone | Approved | [17] | ||
| Synonyms |
Deskar; Esbriet; Pirespa; Pirfenidona; Pirfenidonum; AMR 69; P 2116; AMR-69; F-647; KS-5041; Pirfenidona [INN-Spanish]; Pirfenidone (Deskar); Pirfenidone [USAN:INN]; Pirfenidonum [INN-Latin]; S-7701; Pirfenidone (JAN/USAN/INN); 5-Methyl-1-phenyl-1H-pyridin-2-one; 5-Methyl-1-phenyl-2(1H)-pyridone; 5-Methyl-1-phenyl-2-(1H)-pyridone; 5-methyl-1-phenylpyridin-2(1H)-one; 5-methyl-1-phenylpyridin-2-one
Click to Show/Hide
|
|||
| MOA | Modulator | |||
| External Link | ||||
| A435 | IND submitted | [18] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| TG-C | Phase 3 | [19] | ||
| Synonyms |
3-Thiaglutaryl-Coa; S-[[(2-Hydroxy-2-oxoethyl)thio]acetyl]coenzyme A; (3R,5S,9R)-1-[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)tetrahydrofuran-2-yl]-3,5,9-trihydroxy-8,8-dimethyl-10,14,19-trioxo-2,4,6-trioxa-18,21-dithia-11,15-diaza-3,5-diphosphatricosan-23-oic acid 3,5-dioxide; TGC
Click to Show/Hide
|
|||
| MOA | Modulator | |||
| External Link | ||||
| LY2157299 | Phase 2/3 | [4] | ||
| Synonyms |
Galunisertib; 700874-72-2; LY 2157299; LY-2157299; UNII-3OKH1W5LZE; 4-(2-(6-methylpyridin-2-yl)-5,6-dihydro-4H-pyrrolo[1,2-b]pyrazol-3-yl)quinoline-6-carboxamide; 3OKH1W5LZE; Galunisertib (LY2157299); AK-79916; 4-[5,6-Dihydro-2-(6-methyl-2-pyridinyl)-4H-pyrrolo[1,2-b]pyrazol-3-yl]-6-quinolinecarboxamide; 4-(2-(6-Methylpyridin-2-yl)-5,6-dihydro-4H-pyrrolo-[1,2-b]pyrazol-3-yl)quinoline-6-carboxamide; 4-[2-(6-methylpyridin-2-yl)-4H,5H,6H-pyrrolo[1,2-b]pyrazol-3-yl]quinoline-6-carboxamide
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| Disitertide | Phase 2 | [20] | ||
| Synonyms |
NAFB-001; Disitertide cream (skin disorders), Digna
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| ACE-1334 | Phase 1 | [21] | ||
| MOA | Antagonist | |||
| External Link | ||||
| SRK-181 | Phase 1 | [22] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| ABBV-151 | Phase 1 | [23] | ||
| MOA | Inhibitor | |||
| External Link | ||||
| Mannose phosphate | Discontinued in Phase 2 | [24] | ||
| Synonyms |
SCHEMBL285569; SB17274
Click to Show/Hide
|
|||
| MOA | Modulator | |||
| External Link | ||||
| ART-144 | Investigative | [25] | ||
| Synonyms |
Anti-inflammatory agent (intra-articular/injectable, sustained release, osteoarthritis), Artyx pharmaceuticals
Click to Show/Hide
|
|||
| MOA | Modulator | |||
| External Link | ||||
| JA00: Abortion | 4 Compound(s) Regulating the Disease | Click to Show/Hide the Full List | ||
| Carboprost Tromethamine | Approved | [26] | ||
| Synonyms |
Hemabate; Carboprost Trometanol; Carboprost trometamol; Carboprost tromethamine [USAN]; U 32921E; Hemabate (TN); Prostin / 15M; U-32921E; Prosta-5,13-dien-1-oic acid, 9,11,15-trihydroxy-15-methyl-, (5Z,9-alpha,11-alpha,13E,15S)-, compd. with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1); Prosta-5,13-dien-1-oic acid, 9,11,15-trihydroxy-15-methyl-, (5Z,9-alpha,11-alpha,13E,15S)-,compd. with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1); (15S)-15-Methylprostaglandin F(sub 2alpha) tromethamine; (15S)-15-Methylprostaglandin F2alpha tromethamine; (E)-7-[(1S,3S,5R)-3,5-dihydroxy-2-[(Z,3S)-3-hydroxy-3-methyloct-1-enyl]cyclopentyl]hept-5-enoic acid; (Z)-7-((1R,2R,3R,5S)-3,5-Dihydroxy-2-((E)-(3S)-3-hydroxy-3-methyl-1-octenyl)cyclopentyl)-5-heptenoic acid compound with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1); 15(S)15-Methyl pgf2-alpha tromethaminesalt; 15(S)15-Methyl prostaglandin F2-alpha tromethamine; 15-methylprostaglandin F2alpha-tromethamine
Click to Show/Hide
|
|||
| External Link | ||||
| Dinoprost Tromethamine | Approved | [27] | ||
| Synonyms |
Dinolytic; Ensaprost; Lutalyse; Prostamate; Zinoprost; Panacelan F tromethamine salt; Pronalgon F; Prostalmon F; PGF2alpha THAM; PGF2alpha tromethamine; PROSTAGLANDIN F2ALPHA TRIS SALT; Prostaglandin F2a tromethamine; Prostaglandin F2alpha tham; Prostaglandin F2alpha tromethamine; Prostin F2 alpha; U 14585; Dinoprost tromethamine [USAN:JAN]; Dinoprost, trometamol salt; Enzaprost F compd. with trisamine; Lutalyse (Veterinary); PGF2-alpha tham; PGF2-alpha tris salt; PGF2-alpha tromethamine; PGF2-alpha tromethamine salt; PGF2alpha-Tris; Prostaglandin F(sub 2a) tromethamine; Prostaglandin F-2alpha tromethamine salt; Prostaglandin F2-alpha THAM; Prostaglandin F2-alpha THAM salt; Prostaglandin F2-alpha tromethamine; Prostin F2 alpha (TN); THAM PGF2-alpha; Tromethamine prostaglandin F2-alpha; U-14583E; Dinoprost tromethamine (JAN/USP); Protaglandin F2-alpha-tham salt; U-14,583E; Prosta-5,13-dien-1-oic acid, (5Z,9-alpha,11-alpha,13E,15S)-9,11,15-trihydroxy-, compd. with trimethylolaminomethane; Prosta-5,13-dien-1-oic acid, (5Z,9-alpha,11-alpha,13E,15S)-9,11,15-trihydroxy-,compd. with trimethylolaminomethane; Prosta-5,13-dien-1-oic acid, 9,11,15-trihydroxy-, (5Z,9alpha,11alpha,13E,15S)-, compd. with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1); (5Z,9alpha,11alpha,13E,15S)-9,11,15-Trihydroxyprosta-5,13-dienoic acid tris salt; (E,Z)-(1R,2R,3R,5S)-7-(3,5-Dihydroxy-2-((3S)-(3-hydroxy-1-octenyl))cyclopentyl)-5-heptenoic acid compound with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1); (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(E,3S)-3-hydroxyoct-1-enyl]cyclopentyl]hept-5-enoic acid; 1,3-Propanediol, 2-amino-2-(hydroxymethyl)-, (5Z,9alpha,11alpha,13E,15S)-9,11,15-trihydroxyprosta-5,13-dien-1-oate (salt); 5-Heptenoic acid, 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-, tham; 7-(3,5-Dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-5-heptenoic acid, tromethamine salt
Click to Show/Hide
|
|||
| External Link | ||||
| GLPG0974 | Phase 2 | [28] | ||
| Synonyms |
compound 99 [PMID 25380412]
Click to Show/Hide
|
|||
| External Link | ||||
| Epostane | Investigative | [29] | ||
| Synonyms |
Epostanum [Latin]; Epostano [Spanish]; WIN 32729; AC1L1HB6; Epostane [USAN:BAN:INN]; 4,5-epoxy-3,17-dihydroxy-4,17-dimethylandrost-2-ene-2-carbonitrile (4alpha,5alpha,17beta); Androst-2-ene-2-carbonitrile, 4,5-epoxy-3,17-dihydroxy-4,17-dimethyl-, (4alpha,5alpha,17beta)-; LS-19418; Androst-2-ene-2-carbonitrile, 3,17-dihydroxy-4,17-dimethyl-4,5-epoxy-, (4-alpha,5-alpha,17-beta)-
Click to Show/Hide
|
|||
| External Link | ||||
References