m6A-centered Crosstalk Information
Mechanism of Crosstalk between m6A Modification and Epigenetic Regulation
| Crosstalk ID |
M6ACROT02025
|
[1] | |||
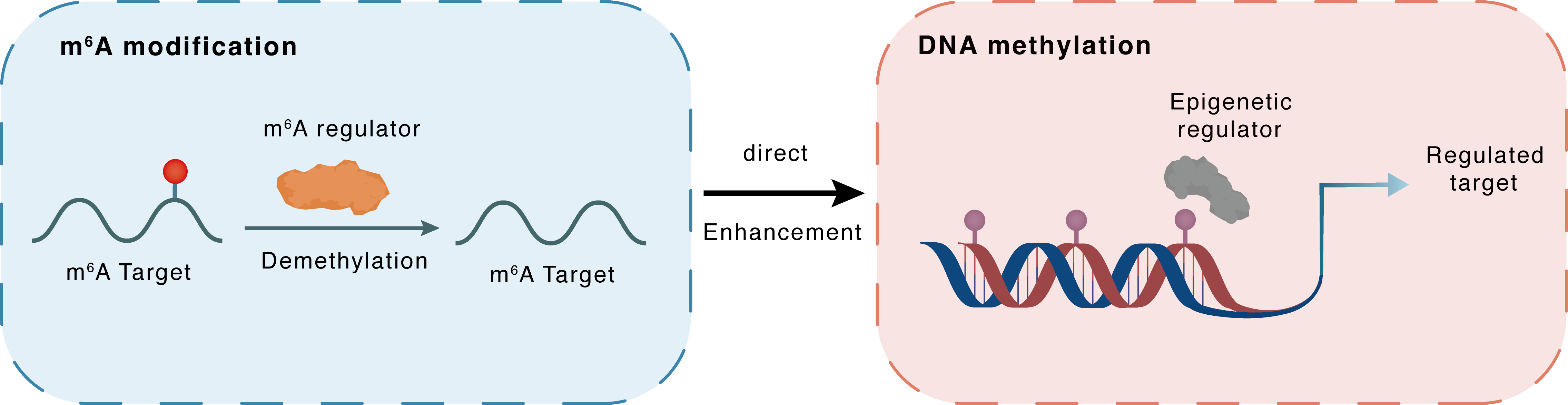 m6A modification
DNMT3B
DNMT3B
ALKBH5
Demethylation
m6A modification
DNMT3B
DNMT3B
ALKBH5
Demethylation
 : m6A sites
Direct
Enhancement
DNA methylation
DNMT3B
E4F1 : m6A sites
Direct
Enhancement
DNA methylation
DNMT3B
E4F1
|
|||||
| m6A Modification: | |||||
|---|---|---|---|---|---|
| m6A Regulator | RNA demethylase ALKBH5 (ALKBH5) | ERASER | |||
| m6A Target | DNA (cytosine-5)-methyltransferase 3B (DNMT3B) | ||||
| Epigenetic Regulation that have Cross-talk with This m6A Modification: | |||||
| Epigenetic Regulation Type | DNA methylation (DNAMeth) | ||||
| Epigenetic Regulator | DNA (cytosine-5)-methyltransferase 3B (DNMT3B) | WRITER | View Details | ||
| Regulated Target | Transcription factor E4F1 (E4F1) | View Details | |||
| Crosstalk Relationship | m6A → DNA methylation | Enhancement | |||
| Crosstalk Mechanism | m6A modification directly impacts DNA methylation through modulating the expression level of DNA methyltransferases or demethylases. | ||||
| Crosstalk Summary | ALKBH5 deficiency-induced m6A hypomethylation of DNA (cytosine-5)-methyltransferase 3B (DNMT3B) leads to less recognition of YTHDF2, resulting in enhanced stability of DNMT3B mRNA. Increased expression of DNMT3B modulated the Transcription factor E4F1 (E4F1) level upon DNA methylation patterns causing cellular arrest and senescent phenotype. | ||||
| Responsed Disease | Intervertebral disc degeneration | ICD-11: FA80 | |||
| Cell Process | Degeneration | ||||
Full List of Potential Compound(s) Related to This m6A-centered Crosstalk
| DNA (cytosine-5)-methyltransferase 3B (DNMT3B) | 22 Compound(s) Regulating the Target | Click to Show/Hide the Full List | ||
| Curcumin | Phase 3 | [2] | ||
| Synonyms |
458-37-7; Diferuloylmethane; Natural yellow 3; Turmeric yellow; Turmeric; Curcuma; Kacha haldi; Gelbwurz; Indian saffron; Curcumin I; Souchet; Halud; Halad; Haidr; Haldar; Merita earth; Yellow Ginger; Terra Merita; Yellow Root; Safran d'Inde; Yo-Kin; Golden seal; Curcuma oil; Orange Root; Oils, curcuma; CI Natural Yellow 3; Curcumine; Hydrastis; Indian turmeric; Yellow puccoon; Turmeric extract; Diferaloylmethane; Kurkumin [Czech]; (1E,6E)-1,7-Bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione; Tumeric yellow; Turmeric oil
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| PMID27376512-Compound-MTC-424 | Patented | [3] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 1940 nM | |||
| External Link | ||||
| PMID27376512-Compound-MTC-427 | Patented | [3] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 295 nM | |||
| External Link | ||||
| PMID27376512-Compound-MTC-422 | Patented | [3] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 1430 nM | |||
| External Link | ||||
| PMID27376512-Compound-MTC-423 | Patented | [3] | ||
| MOA | Inhibitor | |||
| Activity | IC50 = 363 nM | |||
| External Link | ||||
| NSC-622444 | Investigative | [2] | ||
| Synonyms |
NSC622444; CHEMBL116347; AC1Q3LXD; AC1L7GK5; SCHEMBL9755151; dichlorinated diacylmethane fragment; ZINC1616868; BDBM50048522; 5,3'-dicarboxy-4,4'-dihydrodiphenylmethane; 5,5''-methylenebis(3-chloro-2-hydroxybenzoic acid); 5,5'-Methylenebis(3-chloro-2-hydroxybenzoic acid); 3,3'-methanediylbis(5-chloro-6-hydroxybenzoic acid); 5-(3-carboxy-5-chloro-4-hydroxybenzyl)-3-chloro-2-hydroxybenzoic acid; 3',3-Dichloro-4',4-dimethoxy-5',5-bis(methoxycarbonyl)-1,1-diphenylmethane
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-138419 | Investigative | [2] | ||
| Synonyms |
NSC138419; n-[4-(methylamino)benzoyl]glutamic acid; AC1Q5SG8; AC1L5YW4; SCHEMBL5925511; CHEMBL591443; CTK1H0013; 2-[(4-methylaminobenzoyl)amino]pentanedioic acid; A816490; 2-[[4-(methylamino)benzoyl]amino]pentanedioic acid; 2-[[4-(methylamino)phenyl]carbonylamino]pentanedioic acid; 2-[[[4-(methylamino)phenyl]-oxomethyl]amino]pentanedioic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-319745 | Investigative | [2] | ||
| Synonyms |
61629-60-5; HB 093; BRN 2168571; 4-(2-((5-Chloro-2-methoxybenzoyl)amino)ethyl)hydrocinnamic acid; 3-[4-[2-[(5-CHLORO-2-METHOXY-BENZOYL)AMINO]ETHYL]PHENYL]PROPANOIC ACID; 3-(4-(2-(5-Chlor-2-methoxy-benzamido)-aethyl)phenyl)-propionsaeure [German]; 3-[4-[2-[(5-chloro-2-methoxybenzoyl)amino]ethyl]phenyl]propanoic acid; HYDROCINNAMIC ACID, 4-(2-((5-CHLORO-2-METHOXYBENZOYL)AMINO)ETHYL)-; AC1L2AFL; CHEMBL597112; SCHEMBL11481071; CTK5B3505; DTXSID00210642; AIEFQKOARQRACO-UHFFFAOYSA-N; ZINC1572211; HB-093; NSC319745
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-348926 | Investigative | [2] | ||
| Synonyms |
NSC348926; 2-phthalimidoadipic acid; AC1L7IP1; SCHEMBL9741723; CHEMBL599367; 2-(1,3-dioxoisoindol-2-yl)hexanedioic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-401077 | Investigative | [2] | ||
| Synonyms |
NSC401077; MLS000757170; DNA Methyltransferase Inhibitor; CHEMBL383475; 32675-71-1; 2-(1,3-Dioxo-1,3-dihydro-isoindol-2-yl)-3-(1H-indol-3-yl)-propionic acid; 2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)-3-(1H-indol-3-yl)propanoic acid; 2-(1,3-Dioxo-1,3-dihydro-2H-isoindol-2-yl)-3-(1H-indol-3-yl)propanoic acid; 2-(1,3-Dioxo-1,3-dihydro-2H-isoindol-2-yl)-3-(1H-indol-3-yl)propionic acid; SMR000413613; AC1Q71QA; Oprea1_475901; Oprea1_410805; MLS000777218; MLS006011919; SCHEMBL562060
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| S-tubercidinylhomocysteine | Investigative | [4] | ||
| Synonyms |
CHEMBL552309; 57344-98-6; AC1L3YAS; AC1Q5QMO; (S)-7-(5-S-(3-amino-3-carboxypropyl)-5-thio-beta-D-ribofuranosyl)-7H-pyrrolo(2,3-d)pyrimidin-4-amine; (2s)-2-amino-4-({[(2s,3s,4r,5r)-5-(4-amino-7h-pyrrolo[2,3-d]pyrimidin-7-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl}sulfanyl)butanoic acid(non-preferred name); BDBM50294482; (2S)-2-amino-4-[[(2S,3S,4R,5R)-5-(4-aminopyrrolo[2,3-d]pyrimidin-7-yl)-3,4-dihydroxyoxolan-2-yl]methylsulfanyl]butanoic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| Activity | IC50 = 300 nM | |||
| External Link | ||||
| NSC-106084 | Investigative | [2] | ||
| Synonyms |
CHEMBL597113; NSC106084; AC1L6H8Q; CTK7J5419; ZINC1868549; BDBM50308983; {4-[5-bromo-2-(carboxymethoxy)benzoyl]phenoxy}acetic acid; 2-(4-bromo-2-(4-(carboxymethoxy)benzoyl)phenoxy)acetic acid; 2-[4-[5-bromo-2-(carboxymethyloxy)benzoyl]phenoxy]acetic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-154957 | Investigative | [2] | ||
| Synonyms |
NSC154957; AC1L6EF2; CHEMBL586418; 3-benzhydrylsulfanyl-2-formamidopropanoic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-54162 | Investigative | [2] | ||
| Synonyms |
NSC54162; AC1Q5WTA; AC1L6CM2; CHEMBL611994; 2,2'-{[(2-hydroxyphenyl)methanediyl]disulfanediyl}diacetic acid; ZINC1685025; Acetic acid, (salicylidenedithio)di-; 4265-51-4; Acetic acid, [(o-hydroxybenzylidene)dithio]di-; Acetic acid,2'-[[(2-hydroxyphenyl)methylene]bis(thio)]bis-; 2-[carboxymethylsulfanyl-(2-hydroxyphenyl)methyl]sulfanylacetic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-57893 | Investigative | [2] | ||
| Synonyms |
MLS002667915; 7399-94-2; 4-[(1h-benzimidazol-2-ylmethyl)(formyl)amino]benzoic acid; NSC57893; AC1L6GFK; AC1Q5TWY; NCIOpen2_002368; CHEMBL599366; 4-[1H-benzimidazol-2-ylmethyl(formyl)amino]benzoic acid; CTK5D9099; DTXSID30288854; HMS3089M13; ZINC1688755; AKOS030547711
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-56071 | Investigative | [2] | ||
| Synonyms |
32230-52-7; NSC56071; AC1L6EJV; AC1Q7ES0; NCIOpen2_007380; CHEMBL596910; CTK4G8394; DTXSID80288485; ZINC1686711; 2,2'-[piperazine-1,4-diylbis(carbonothioylsulfanediyl)]diacetic acid; AKOS030574801; Acetic acid,2,2'-[1,4-piperazinediylbis(carbonothioylthio)]bis- (9CI); 2-[4-(carboxymethylsulfanylcarbothioyl)piperazine-1-carbothioyl]sulfanylacetic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-137546 | Investigative | [2] | ||
| Synonyms |
NSC137546; CHEMBL591202; AC1L5Y49; AKOS008984447; 2-[(2,6-dichlorobenzoyl)amino]pentanedioic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-623548 | Investigative | [2] | ||
| Synonyms |
2581-36-4; NSC 408488; o-Cresotic acid, 5,5'-methylenedi-; 2,3-CRESOTIC ACID, 5,5'-METHYLENEDI-; UNII-S3D8KC88KC; 5,5'-Methylenedi-2,3-cresotic acid; NSC 623548; BRN 3433298; S3D8KC88KC; CHEMBL113835; 5,5'-Methylenedi-o-cresotic acid; NSC623548; NSC408488; 5,5'-Methylenebis(2-hydroxy-3-methylbenzoic acid); 2, 5,5'-methylenedi-; AC1L29YK; Oprea1_231968; 2-10-00-00398 (Beilstein Handbook Reference); SCHEMBL9755153; CTK4F6504; DTXSID90180466; o-Cresotic acid,5'-methylenedi-; MolPort-000-698-522; ZINC4028795; STL511095
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-345763 | Investigative | [2] | ||
| Synonyms |
7-(8-hydroxyquinolin-5-yl)-4,7-dioxoheptanoic acid; NSC345763; AC1L7HSU; CHEMBL597114
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| NSC-158324 | Investigative | [2] | ||
| Synonyms |
Acediasulfone; UNII-30YP2YHH8W; 30YP2YHH8W; CHEMBL48396; N-[4-[(4-AMINOPHENYL)SULPHONYL]PHENYL]GLYCINE; 2-[4-(4-aminophenyl)sulfonylanilino]acetic acid; Acediasulfonum; N-(4-((4-Aminophenyl)sulphonyl)phenyl)glycine; EINECS 201-243-7; AC1L25EF; ZINC862; SCHEMBL143660; CTK5E7379; DTXSID00229991; CHEBI:135300; BDBM50099670; AKOS027327086; DB08926; Glycine,N-[4-[(4-aminophenyl)sulfonyl]phenyl]-; {4-[(4-aminophenyl)sulfonyl]anilino}acetic acid; 2-(4-(4-aminophenylsulfonyl)phenylamino)acetic acid
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| External Link | ||||
| (L-)-S-adenosyl-L-homocysteine | Investigative | [5] | ||
| Synonyms |
S-Adenosyl-L-homocysteine; S-adenosylhomocysteine; 979-92-0; AdoHcy; S-(5'-adenosyl)-L-homocysteine; adenosylhomocysteine; Formycinylhomocysteine; Adenosyl-L-homocysteine; S-(5'-deoxyadenosin-5'-yl)-L-homocysteine; 2-S-adenosyl-L-homocysteine; 5'-Deoxy-S-adenosyl-L-homocysteine; S-adenosyl-homocysteine; S-Adenosyl Homocysteine; L-S-Adenosylhomocysteine; L-Homocysteine, S-(5'-deoxyadenosin-5'-yl)-; adenosylhomo-cys; adenosyl-homo-cys; UNII-8K31Q2S66S; (S)-5'-(S)-(3-Amino-3-carboxypropyl)-5'-thioadenosine; BRN 5166233; SAH
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| Activity | IC50 = 200 nM | |||
| External Link | ||||
| NSC-622445 | Investigative | [2] | ||
| Synonyms |
5,5'-Methylenedisalicylic acid; 122-25-8; 5,5'-Methylenebis(2-hydroxybenzoic acid); Methylenebis(salicylic acid); 5,5-Methylenebis(salicylic acid); UNII-2KF4FVV76N; 5,5-Methylenedisalicylic acid; 5-(3-Carboxy-4-hydroxybenzyl)salicylic acid; 4,4'-Dihydroxy-3,3'-dicarboxydiphenylmethane; 3,3'-Dicarboxy-4,4'-dihydroxydiphenylmethane; NSC 14778; 2KF4FVV76N; 4,4'-Dihydroxydiphenylmethane-3,3'-dicarboxylic acid; 3,3'-Methylenebis(6-hydroxybenzoic acid); CHEMBL115145; Benzoic acid, 3,3'-methylenebis[6-hydroxy-
Click to Show/Hide
|
|||
| MOA | Inhibitor | |||
| Activity | IC50 = 17000 nM | |||
| External Link | ||||
| FA80: Intervertebral disc degeneration | 3 Compound(s) Regulating the Disease | Click to Show/Hide the Full List | ||
| Rexlemestrocel-L | Phase 3 | [6] | ||
| Synonyms |
MPC-150-M
Click to Show/Hide
|
|||
| External Link | ||||
| CybroCell | Phase 1/2 | [7] | ||
| External Link | ||||
| IDCT | Phase 1/2 | [8] | ||
| Synonyms |
rebonuputemcel
Click to Show/Hide
|
|||
| External Link | ||||
References