m6A-centered Drug Response Information
General Information of the Drug (ID: M6APDG03324)
| Name |
BNP-1350
|
||||
|---|---|---|---|---|---|
| Synonyms |
Cositecan; Cositecan (USAN); Cositecan [USAN:INN]; Karenitecin; Karenitecin (TN); POADTFBBIXOWFJ-VWLOTQADSA-N; SCHEMBL2315201; Z-3161; ZINC169746728; 1H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione, 4-ethyl-4-hydroxy-11-(2-trimethylsilyl)ethyl)-, (4S)-; 203923-89-1; 24R60NVC41; 7-Trimethylsilylethylcamptothecin; 923K891; AC1L3WN8; AN-29989; BNP 1350; BNP-1350; BNP1350; CHEMBL1997373; CS-0003573; D09327; DB 172; DB05806; DTXSID90174340; HY-14812; NCI60_038797; NSC-710270; NSC710270; UNII-24R60NVC41
Click to Show/Hide
|
||||
| Status |
Phase 3
|
||||
| Structure |
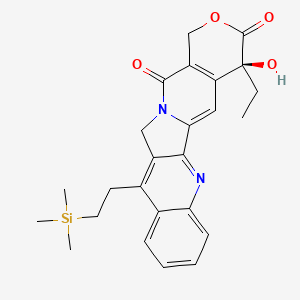 |
||||
| Formula |
C25H28N2O4Si
|
||||
| InChI |
1S/C25H28N2O4Si/c1-5-25(30)19-12-21-22-17(13-27(21)23(28)18(19)14-31-24(25)29)15(10-11-32(2,3)4)16-8-6-7-9-20(16)26-22/h6-9,12,30H,5,10-11,13-14H2,1-4H3/t25-/m0/s1
|
||||
| InChIKey |
POADTFBBIXOWFJ-VWLOTQADSA-N
|
||||
| PubChem CID | |||||
| VARIDT Drug ID | |||||
| INTEDE Drug ID | |||||
Target Gene(s) and Their Upstream m6A Regulator, Together with the Effect of Target Gene(s) in Drug Response
The target genes involved in drug-target interaction (such as drug-metabolizing enzymes, drug transporters and therapeutic targets) and drug-mediated cell death signaling (including modulating DNA damage and repair capacity, escaping from drug-induced apoptosis, autophagy, cellular metabolic reprogramming, oncogenic bypass signaling, cell microenvironment, cell stemness, etc.) could be regulated by m6A regulator(s) and affected their corresponding drug response. You can browse detailed information on drug-related target gene(s) mediated by m6A regulators.
Breast cancer resistance protein (ABCG2)
Methyltransferase-like 3 (METTL3)
| In total 1 mechanisms lead to this potential drug response | ||||
| Response Summary | Breast cancer resistance protein (ABCG2) is a therapeutic target for BNP-1350. The Methyltransferase-like 3 (METTL3) has potential in affecting the response of BNP-1350 through regulating the expression of Breast cancer resistance protein (ABCG2). | [1], [2] | ||
Cytochrome P450 2C8 (CYP2C8)
Fat mass and obesity-associated protein (FTO)
| In total 1 mechanisms lead to this potential drug response | ||||
| Response Summary | Cytochrome P450 2C8 (CYP2C8) is a therapeutic target for BNP-1350. The Fat mass and obesity-associated protein (FTO) has potential in affecting the response of BNP-1350 through regulating the expression of Cytochrome P450 2C8 (CYP2C8). | [3], [4] | ||
Methyltransferase-like 14 (METTL14)
| In total 1 mechanisms lead to this potential drug response | ||||
| Response Summary | Cytochrome P450 2C8 (CYP2C8) is a therapeutic target for BNP-1350. The Methyltransferase-like 14 (METTL14) has potential in affecting the response of BNP-1350 through regulating the expression of Cytochrome P450 2C8 (CYP2C8). | [3], [4] | ||
Methyltransferase-like 3 (METTL3)
| In total 1 mechanisms lead to this potential drug response | ||||
| Response Summary | Cytochrome P450 2C8 (CYP2C8) is a therapeutic target for BNP-1350. The Methyltransferase-like 3 (METTL3) has potential in affecting the response of BNP-1350 through regulating the expression of Cytochrome P450 2C8 (CYP2C8). | [3], [4] | ||
YTH domain-containing protein 2 (YTHDC2)
| In total 1 mechanisms lead to this potential drug response | ||||
| Response Summary | Cytochrome P450 2C8 (CYP2C8) is a therapeutic target for BNP-1350. The YTH domain-containing protein 2 (YTHDC2) has potential in affecting the response of BNP-1350 through regulating the expression of Cytochrome P450 2C8 (CYP2C8). | [3], [4] | ||
References