m6A-centered Crosstalk Information
Mechanism of Crosstalk between m6A Modification and Epigenetic Regulation
| Crosstalk ID |
M6ACROT06010
|
[1] | |||
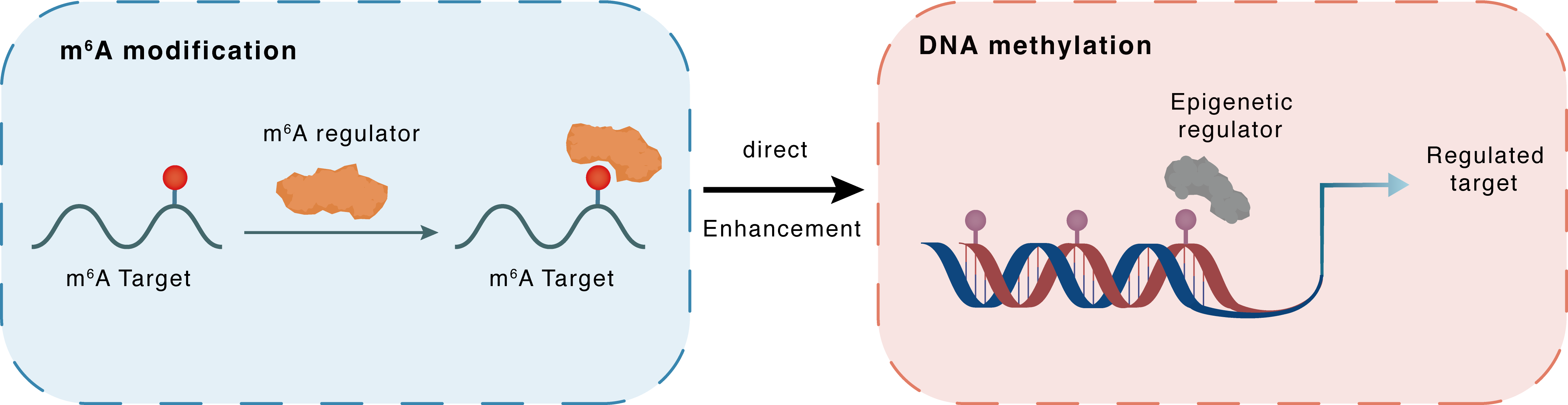 m6A modification
FAT4
FAT4
FXR1
m6A modification
FAT4
FAT4
FXR1
 : m6A sites
Direct
Enhancement
DNA methylation
TET1
FAT4 : m6A sites
Direct
Enhancement
DNA methylation
TET1
FAT4
|
|||||
| m6A Modification: | |||||
|---|---|---|---|---|---|
| m6A Regulator | RNA-binding protein FXR1 (FXR1) | READER | |||
| m6A Target | Protocadherin Fat 4 (FAT4) | ||||
| Epigenetic Regulation that have Cross-talk with This m6A Modification: | |||||
| Epigenetic Regulation Type | DNA methylation (DNAMeth) | ||||
| Epigenetic Regulator | Methylcytosine dioxygenase TET1 (TET1) | ERASER | View Details | ||
| Regulated Target | Protocadherin Fat 4 (FAT4) | View Details | |||
| Crosstalk Relationship | m6A → DNA methylation | Enhancement | |||
| Crosstalk Mechanism | m6A modification directly impacts DNA methylation through recruiting DNA methyltransferases or demethylases. | ||||
| Crosstalk Summary | Here, we show that METTL3-mediated RNA N6-methyladenosine (m6A) formation leads to DNA demethylation in nearby genomic loci in normal and cancer cells, which is mediated by the interaction between m6A reader FXR1 and DNA 5-methylcytosine dioxygenase TET1. Upon recognizing RNA m6A, FXR1 recruits TET1 to genomic loci to demethylate DNA, leading to reprogrammed chromatin accessibility and gene transcription. These gene include SATB2, RPIA, WNT7B, BCL6, Protocadherin Fat 4 (FAT4) and SAMD9L. | ||||
| Responsed Disease | Esophageal Squamous Cell Carcinoma | ICD-11: 2B70.1 | |||
| Cell Process | Transcription | ||||
| Cell proliferation | |||||
| Cell migration | |||||
| Cell invasion | |||||
In-vitro Model |
HEK293T | Normal | Homo sapiens | CVCL_0063 | |
| KYSE-30 | Esophageal squamous cell carcinoma | Homo sapiens | CVCL_1351 | ||
| KYSE-510 | Esophageal squamous cell carcinoma | Homo sapiens | CVCL_1354 | ||
Full List of Potential Compound(s) Related to This m6A-centered Crosstalk
| 2B70: Esophageal cancer | 15 Compound(s) Regulating the Disease | Click to Show/Hide the Full List | ||
| Pembrolizumab | Approved | [2] | ||
| External Link | ||||
| Nivolumab | Approved | [2] | ||
| External Link | ||||
| Claudiximab | Phase 3 | [3] | ||
| Synonyms |
IMAB-362; Anti-GC182 mAbs (cancer), Ganymed; Anti-GC182 monoclonal antibodies (cancer), Ganymed; Anti-CLDN18-2 mAbs (cancer), Ganymed; Anti-CLDN18-2 monoclonal antibodies (cancer), Ganymed
Click to Show/Hide
|
|||
| External Link | ||||
| Golnerminogene pradenovac | Phase 3 | [4] | ||
| Synonyms |
TNFerade (TN)
Click to Show/Hide
|
|||
| External Link | ||||
| DKN-01 | Phase 2 | [5] | ||
| External Link | ||||
| Pegamotecan | Phase 2 | [6] | ||
| Synonyms |
Prothecan; EZ-246; PEG-camptothecin; PEG-camptothecin, Enzon; Polyethylene glycol-camptothecin, Enzon
Click to Show/Hide
|
|||
| External Link | ||||
| APR-246 | Phase 2 | [2] | ||
| Synonyms |
Eprenetapopt
Click to Show/Hide
|
|||
| External Link | ||||
| Opdivo + Yervoy | Phase 3 | [2] | ||
| External Link | ||||
| Anti-NY-ESO-1 CAR-T cells | Phase 1/2 | [7] | ||
| External Link | ||||
| Anti-MUC1 CAR-T cells | Phase 1/2 | [8] | ||
| External Link | ||||
| CAR-T Cells targeting EpCAM | Phase 1/2 | [9] | ||
| External Link | ||||
| PCA062 | Phase 1 | [2] | ||
| External Link | ||||
| Cellspan esophageal implant | Clinical trial | [2] | ||
| External Link | ||||
| PKI166 | Discontinued in Phase 2 | [10] | ||
| Synonyms |
PKI-166; CGP-75166; 187724-61-4; NVP-PKI166; CHEMBL1914653; AC1OCFE0; UNII-9RIE5HW38P; 9RIE5HW38P; SCHEMBL177814; GTPL7642; CHEMBL1963502; ZINC23255; AOB1619; PKI-75166; BDBM50358046; NCGC00387215-02; AS-16676; KB-275097; PKI-166, > 4-[4-[[(1R)-1-phenylethyl]amino]-7H-pyrrolo[4,5-e]pyrimidin-6-yl]phenol
Click to Show/Hide
|
|||
| External Link | ||||
| Ramorelix | Discontinued in Phase 1 | [11] | ||
| Synonyms |
Hoe-013
Click to Show/Hide
|
|||
| External Link | ||||
References